In the beginning there were SMA and DIBMA.
But now, a new member joins the club of synthetic polymer nanodiscs:
AASTY - Acrylic Acid Co-Styrene
Around one-third of known human protein-coding genes are coding for secreted or membrane-bound proteins. Although this is a significant portion of the whole proteome, only a few of them are structurally known. As about half of all approved therapeutics target membrane proteins, an improvement of this status is highly favorable for future drug design (Baker, 2010). This is where synthetic polymer nanodiscs such as AASTY come in.
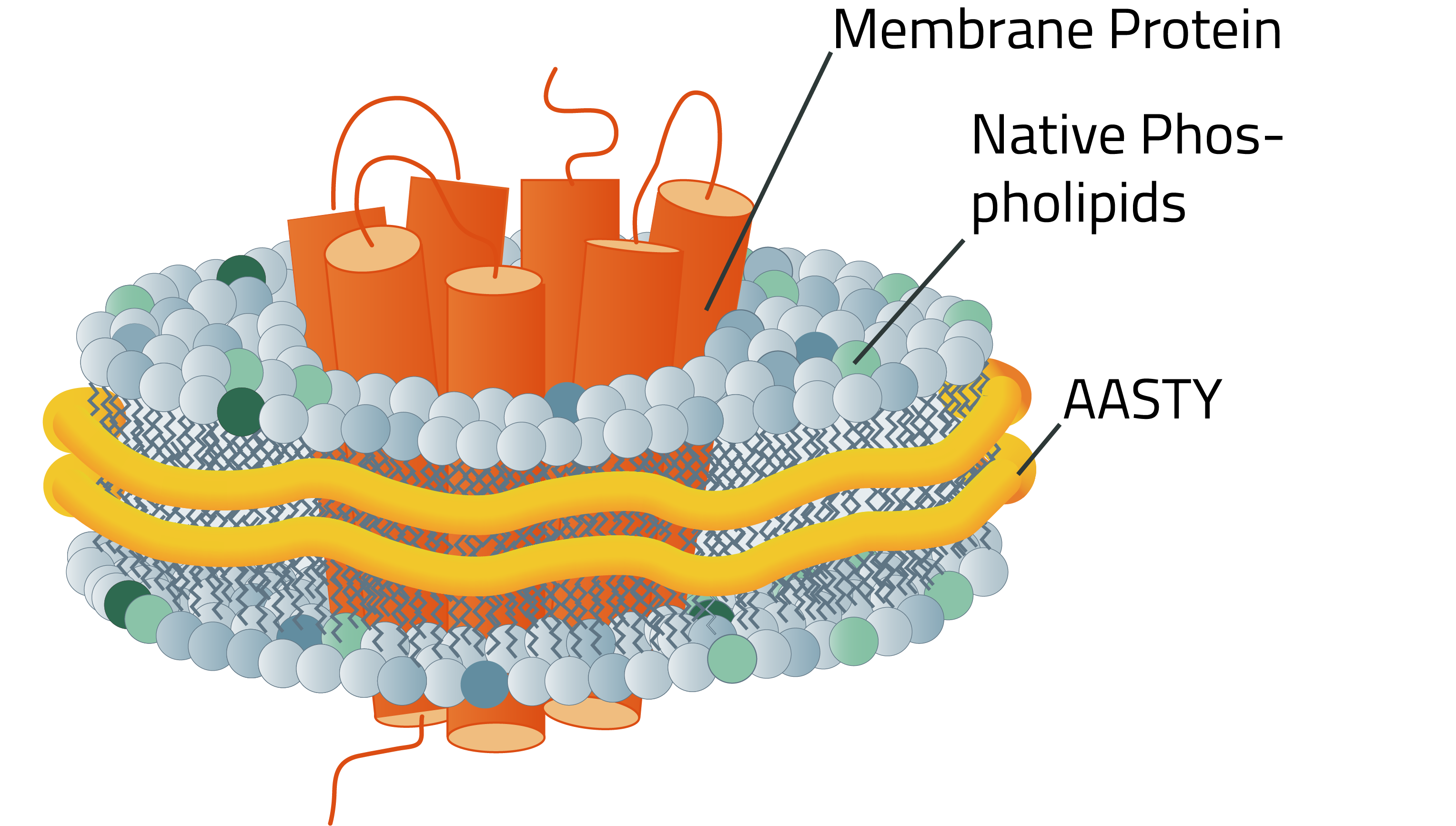
What is AASTY?
Poly(acrylic acid-co-styrene), AASTY, or SAA, is a highly-alternating copolymer composed of acrylic acid and styrene.
AASTY is well-suited for native lipid nanodiscs and effective in the extraction of membrane proteins from mammalian, bacterial, and yeast systems (1,2). The use of AASTY for native nanodisc preparations was developed by Dr. Anton Autzen and Dr. Henriette Autzen, in collaboration with the Appel Research Group at Stanford University, and the Yifan Cheng Lab at University of California San Francisco.
Why should you use AASTY?
Why should you use AASTY?The reactivity ratio of styrene and acrylic acid, allows for a highly alternating monomer sequence in the copolymer (1). This means that they are more homogeneous and consequently more efficient than copolymers that are less homogeneous. The exact composition of AASTY copolymers show different extraction efficiencies, depending on the lipid composition of the lipid bilayers being formulated into nanodiscs.
Excess AASTY copolymer can be removed by dialysis after nanodisc formation (3). This is because the dispersity of the polymers molecular weight distribution is low and the molecular weight is highly controlled in the reversible addition−fragmentation chain-transfer (RAFT) reaction used to synthesize AASTY. The illustration to the right highlights this aspect.
Based on previous findings on SMA, it is the expectation that AASTYs of different molecular weights will display different rates of nanodisc formation, extraction efficacy, and stability of resulting nanodiscs.
Excess AASTY copolymer can be removed by dialysis after nanodisc formation (3). This is because the dispersity of the polymers molecular weight distribution is low and the molecular weight is highly controlled in the reversible addition−fragmentation chain-transfer (RAFT) reaction used to synthesize AASTY. The illustration to the right highlights this aspect.
Based on previous findings on SMA, it is the expectation that AASTYs of different molecular weights will display different rates of nanodisc formation, extraction efficacy, and stability of resulting nanodiscs.

Features - Our Individual AASTYs in comparison
Things to know before:
- Ratio of Acrylic Acid to Styrene - 45:55, 50:50 or 55:45 >> Acrylic Acid contributes to hydrophilic property of AASTY
- 5-6 kDa more aggressive; 10-11 kDa thermodynamic stable >> Acrylic Acid contributes to charge
| Name | Molecular Weight | pH after solving | Divalent cationic tolerance | Acrylic Acid : Styrene Ratio |
|---|---|---|---|---|
| AASTY 6-45 | 5-6 kDa | 7.5 | 6 mM | 45% to 55% |
| AASTY 6-50 | 5-6 kDa | 7.5 | 6 mM | 50% to 50% |
| AASTY 6-55 | 5-6 kDa | 7.5 | 6 mM | 55% to 45% |
| AASTY 11-45 | 10-11 kDa | 7.5 | 6 mM | 45% to 55% |
| AASTY 11-50 | 10-11 kDa | 7.5 | 6 mM | 50% to 50% |
| AASTY 11-55 | 10-11 kDa | 7.5 | 6 mM | 55% to 45% |
The purpose of nanodiscs
To summarize: Reasons for working with AASTYs instead of detergents!



in native-like conditions.


