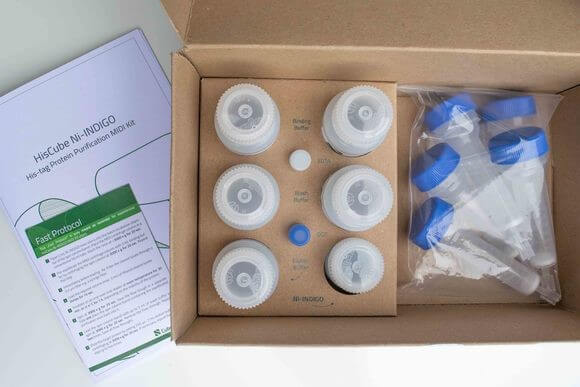
Ni-INDIGO
Cube Biotech introduces the novel Ni-INDIGO product line for His-tag protein purifications. The novel ligand INDIGO is loaded with nickel ions. Using the INDIGO ligand, purification of His-tagged proteins is possible in the presence of up to 20 mM DTT and 20 mM EDTA at high protein capacity. Please note that the novel INDIGO ligand cannot be stripped with EDTA.
Note: Due to increased requests the regeneration protocol for all INDIGO products can be found here.
-
Products
- Membrane Protein Stabilization
- Protein Purification Products
- Active Proteins
- Proteomics
- Cell-Free Lysates
- Plastic & Accessories
Filter
–