Introduction To Agarose Matrices For Protein Purification
What are Agarose Beads?
Agarose beads are hydrogels with different particle diameters and concentrations. Agarose is made of repeating units of agarobiose. Agarose beads are normally cross-linked and porous so protein may flow through the beads. The beads are useful for size exclusion or affinity chromatography. For affinity chromatography ligands (like NTA or IDA) are covalently linked to the agarose bead polymer so tagged proteins can be separated from other proteins.
Agarose matrices have been used for protein purification for decades, and a broad variety of agarose-based matrices is commercially available. Physical properties can vary significantly between the different matrices. This short summary shall support you in finding the optimal product for your research.
Agarose matrices have been used for protein purification for decades, and a broad variety of agarose-based matrices is commercially available. Physical properties can vary significantly between the different matrices. This short summary shall support you in finding the optimal product for your research.
Superflow, Sepharose, Agarose - what's the difference?
In the past 20+ years, many developments have been made, and names have been created for different kinds of agarose matrices:
PureCube Agarose by Cube Biotech
Our PureCube Agarose is highly-crosslinked and pressure-stable and therefore comparable to Sepharose and Superflow matrices (see Table 1). For high flow rates and uncompromised protein binding capacity, we recommend our novel PureCube 100 Agarose, offered for His affinity purification.
Sepharose
GE trademark for crosslinked agarose in different concentrations and sizes. Properties of Sepharose vary depending on agarose concentration and degree of cross-linking, and not all Sepharose matrices are suitable for applications such as FPLC.
Superflow
Offered by several companies. The term describes a variant of agarose that is highly cross-linked and pressure-stable, thereby suitable for FPLC experiments. This term is mostly used to distinguish between pressure-stable agaroses and others that show a lower degree of cross-linking and are mostly used for batch purifications only.
Table.1: Comparison of three commercially available agarose matrices.
| Cube Biotech | Supplier G "Sepharose" | Supplier Q "Superflow" | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Material | Highly cross-linked agarose | Highly cross-linked agarose | Highly cross-linked agarose | ||||||
| Agarose concentration |
|
6% / 4% | 6% | ||||||
| Pore size | medium | medium | medium | ||||||
| Mean particle size |
|
34 µm (HP) 90 µm (FF) | 60-160 µm | ||||||
| Recommended linear flow rate | >500 cm/h (PureCube) >1,000 cm/h (PureCube 100) |
>150 cm/h | >150 cm/h | ||||||
| Max. flow rate | 6 ml/min | 4 or 20 ml/min (1 or 5 ml column) |
20 ml/min | ||||||
| Binding capacity: His-tagged proteins and Ni-NTA | up to 80 mg | 40 mg | up to 50 mg |
Kindly provided by PAIA Biotech, Cologne, Germany
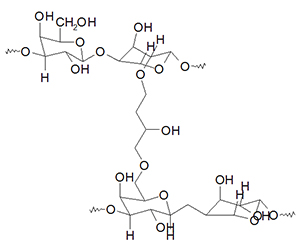
Pore size
The higher the initial agarose concentration, the smaller the pore sizes that form after cross-linking. This is particularly important if the agarose is used for size exclusion chromatography.
Pressure stability
The higher the initial agarose concentration, the higher the degree of crosslinking, and the more pressure-resistant the beads become.
Pressure stability
The higher the initial agarose concentration, the higher the degree of crosslinking, and the more pressure-resistant the beads become.
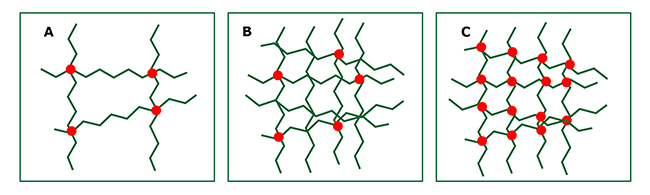
- low concentrated agarose
- high concentrated agarose with a low degree of crosslinking
- high concentrated agarose with a high degree of crosslinking
Why do proteins bind to agarose beads?
Agarose beads bind naturally not themselves to proteins. They have to be attached to ligands such as NTA, glutathione, or antibodies, to enable the purification of proteins via a specific interaction. For affinity chromatography, the most used agarose beads are Nickel-NTA beads.
Ion exchange chromatography
Functional groups such as quarternary amines, DEAE, sulfopropyl, or carboxymethyl can be coupled to the agarose, leading to strong or weak anion exchangers, or strong or weak cation exchangers, respectively. For ion exchange chromatography, proteins do not require an affinity tag, making the method suitable for the purification of proteins from natural sources.
Gel filtration | Size exclusion chromatography
Crosslinked agaroses can be used for size-exclusion chromatography without further modification. Resolution and size range depends on pore sizes. As a rule of thumb, the correlation of agarose concentration to separation capability is as follows:
Table 2: Relationship of agarose concentration to protein exclusion limits
| Agarose concentration | Protein exclusion limit (globular proteins) |
|---|---|
| 4% | ca. 30,000 kDa |
| 5% | ca. 10,000 kDa |
| 6% | ca. 4,000 kDa |
| 7,5% | ca. 1,200 kDa |
| 9% | ca. 150 kDa |
Different bead sizes - is there a reason for it?
Frequently asked questions in terms of agarose resins and magnetic agarose beads are:
- "What are these different sizes good for?"
- "What bead size fits my needs the best?"
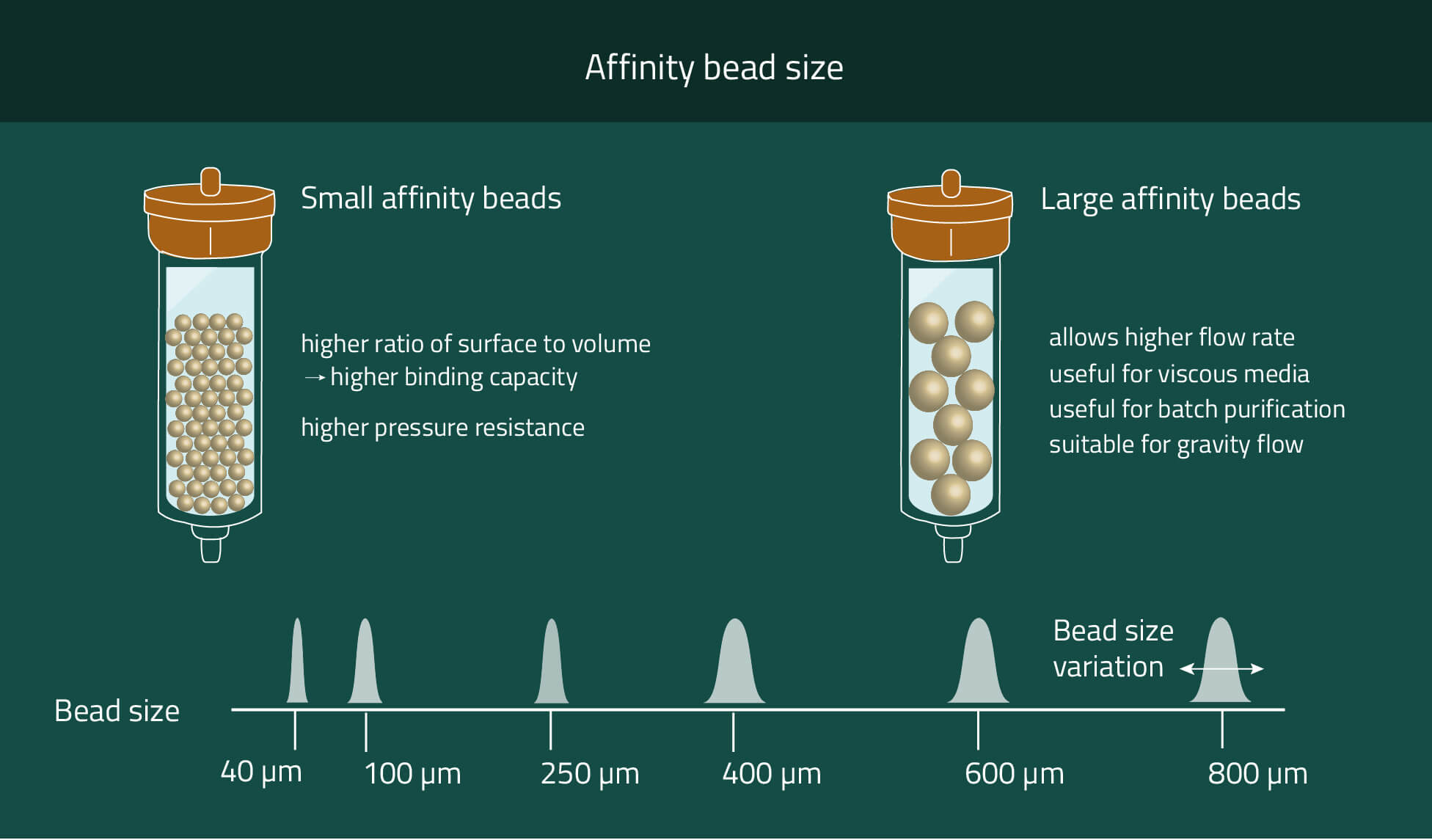
Pressure
The smaller the beads, and the more narrow the size distribution, the higher the pressure-resistance of the beads. Oppositely, the larger the beads, the faster the flow rate in batch and FPLC experiments.
Binding capacity
The smaller the beads, the higher the ratio of surface to volume, and the higher the binding capacity of the beads. However, note that the binding capacity also depends on other factors such as the type and size of the ligand and the density of ligand coupling.
Dynamic binding capacity
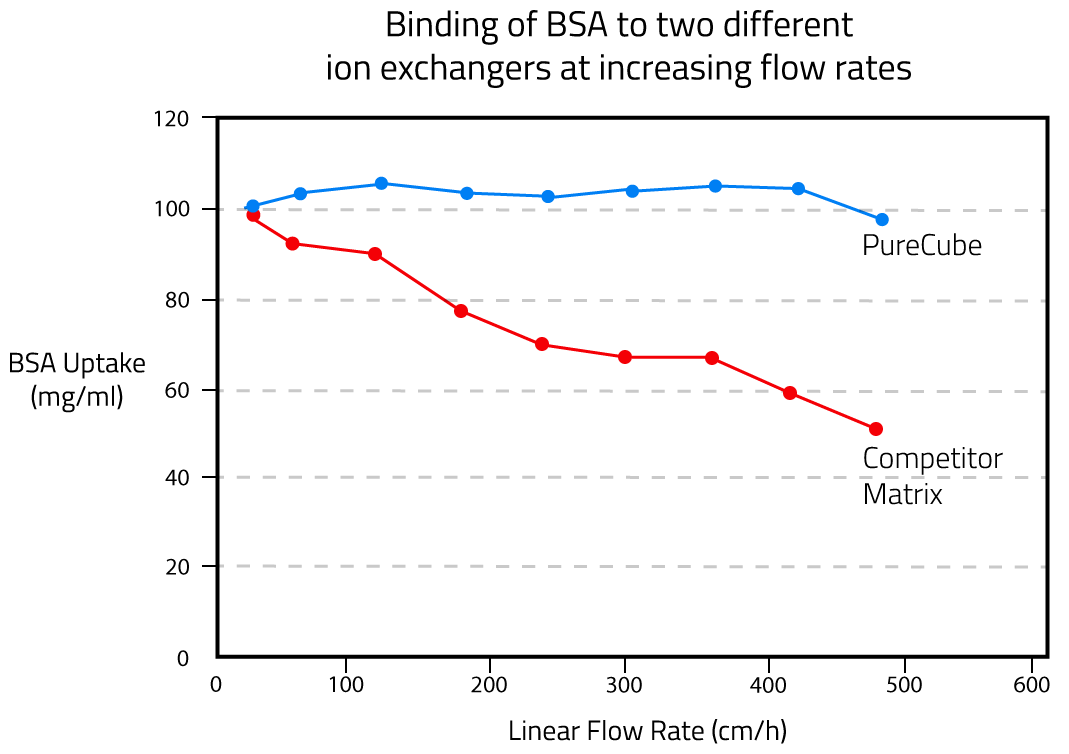
The binding capacity of affinity chromatography material decreases when pressure builds up inside the column at higher flow rates. This effect depends on agarose bead size, agarose content, and degree of crosslinking. The dynamic binding capacity tells us whether an agarose material can be used at higher flow rates without influencing protein yields in a purification. See Fig. 5 for a comparison of PureCube Agarose and a competing product.
Cube Biotech's standard bead sizes
At Cube Biotech, we are able to create agarose resin beads of various sizes to fit your specific needs. However, we also have our standard sizes that can be used for most of our products.Agarose resin beads come in three standard sizes in the Cube Biotech shop: 40µm, 100µm, and 400µm (XL).
MagBeads are available in two standard sizes: 30µm and 90µm (XL).




