PureCube Co-NTA Agarose
Order number: 31403
Description
Our PureCube Co-NTA agarose resins are small agarose resin beads with a diameter of 40 µm. They are used for the purification of active His-tagged proteins from cells. Our year-long experience in manufacturing agarose resin lead to the high yield of 40 mg protein per ml resin, which is leading in the market compared to other Co-NTA suppliers. PureCube Co-NTA resins are suited for batch spin columns and FPLC. Their small diameter provides them with great mechanical stability.
For higher flow rates we recommend our Co-NTA agarose beads with 100 µm diameter and for extreme cases our XL-sized Co-NTA beads.
Furthermore, you can also get these beads pre-packed in a column/cartridge or as magnetic beads.
For higher flow rates we recommend our Co-NTA agarose beads with 100 µm diameter and for extreme cases our XL-sized Co-NTA beads.
Furthermore, you can also get these beads pre-packed in a column/cartridge or as magnetic beads.
Datasheets
- Co-NTA agarose Datasheet
- Native Purification Protocol
- Denaturing Purification Protocol
- Batchspin MINI Native Purification Protocol
- Batchspin MINI Denaturing Purification Protocol
- Batchspin MIDI Native Purification Protocol
- Batchspin MIDI Denaturing Purification Protocol
- Washing & Regeneration Protocol
- Cartridge Packing Protocol
| Feature | |
|---|---|
| Usage | Specific binding and purification of 6x His tagged proteins |
| Specificity | Affinity to His tagged proteins |
| Binding capacity | >30 mg/mL |
| Bead Ligand | Co-NTA |
| Bead size | 40 μm |
| Filling quantity | Delivered as a 50 % suspension |
| Required equipment |
|
Citations
| Purified Protein | Year | Author |
|---|---|---|
| eGFP | 2015 | Li Y., Lui Y., Gao T., Zhang B., Song Y., Terrell J-L., Barber N., Bentley W-E., Takeuchi I., Payne G-F., Wang Q. |
| HrpII | 2017 | Bauer W.S., Richardson K.A., Adams N.M., Ricks K.M., Gasperino D.J., Ghionea S.J., Rosen M., Nichols K.P., Weigl B.H., Haselton F.R., Wright D.W. |
| Flavin-dependent tryptophan 6-halogenase Thal | 2018 | Moritzer A.-C., Minges H., Prior T., Frese M., Seewald N., Niemann H.H. |
| Metal Binding Phages | 2019 | Matys S., Schönberger N., Lederer F., Pollmann K. |
| Flavin-dependent halogenase AetF | 2023 | Gäfe S. & Niemann H. H. |
Lab Results
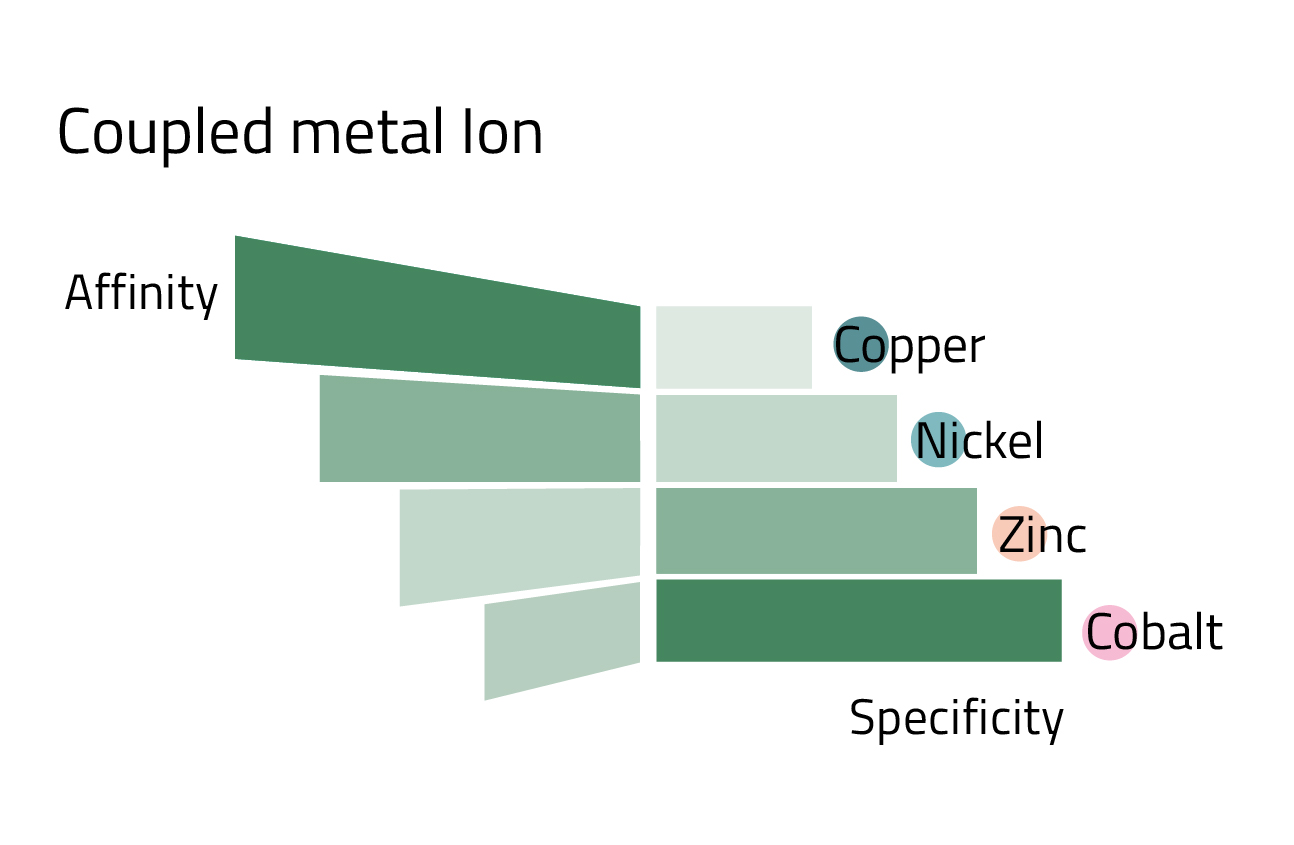
Different metal ions confer different binding affinity and specificity
Loading different metal ions to a resin results in differing affinity and specificity for a His-tagged protein. Generally, cobalt exhibits the highest binding specificity of commonly used IMAC metal ions, leading to relatively low yields but high purity. Copper, at the other end of the spectrum, has a high affinity leading to high yields but unspecific binding. In searching for the optimal resin to purify a protein, it is recommended to explore different chelating ligands (IDA or NTA) and different metal ions.
Loading different metal ions to a resin results in differing affinity and specificity for a His-tagged protein. Generally, cobalt exhibits the highest binding specificity of commonly used IMAC metal ions, leading to relatively low yields but high purity. Copper, at the other end of the spectrum, has a high affinity leading to high yields but unspecific binding. In searching for the optimal resin to purify a protein, it is recommended to explore different chelating ligands (IDA or NTA) and different metal ions.
Video
Video Guide - How to pack FPLC cartridges
Video Guide - FPLC
Video Guide - Column Chromatography
Video Guide - Batch Spin Chromatography
FAQ
Can I get the datasheet for the Co-NTA resin?
Why should I use Co-NTA beads instead of Ni-NTA?
As figure 1 in the "lab results" section illustrates, Co-NTA purifications using the His-tag are purer than Ni-NTA in comparison. This comes at a cost of some of the protein yield. If your needs fit that pattern use Co-NTA beads instead of Ni-NTA.
What are the reasons for non-specific binding?
Even though Co-NTA is the purest form of His-tag purification, some unwanted proteins may still bind to the beads. But washing with NaOH after elution of your protein of interest removes unspecific bound proteins from your resin.
I want to use a high concentration of EDTA and DTT. Is it possible to use Co-NTA from Cube Biotech?
No, it is not recommended because cobalt-ions are reduced with DTT or dissolved with EDTA. If you want to use high concentrations of EDTA and DTT you should use our Indigo resin.
How is the capacity at high flow rates?
If higher flow rates are desired we recommend using beads with bigger diameters. We offer Co-NTA beads with mean diameters of 40µm, 100µm, and 400µm (XL).
With each size increase, the flow rates also increase due to the proportionally increasing space between the beads. However, the surface of the beads does not increase at the same speed as the diameter (square-cube-law). That results in decreasing amounts of purified protein per mL beads while increasing the bead sizes.
For 40µm of 100µm beads, we both have average purification amounts of ~30 µg protein/mL beads. With 400 µm (XL) beads, this decreases to an unspecified amount.
We recommend reading the corresponding section of the "Introduction to agarose matrixes" guide on this subject for more detailed information.
With each size increase, the flow rates also increase due to the proportionally increasing space between the beads. However, the surface of the beads does not increase at the same speed as the diameter (square-cube-law). That results in decreasing amounts of purified protein per mL beads while increasing the bead sizes.
For 40µm of 100µm beads, we both have average purification amounts of ~30 µg protein/mL beads. With 400 µm (XL) beads, this decreases to an unspecified amount.
We recommend reading the corresponding section of the "Introduction to agarose matrixes" guide on this subject for more detailed information.
After using DTT my resin changed color. How to regenerate it?
The DTT has probably destroyed your beads. Co-NTA beads only have a limited DTT tolerance of about 1 mM. However, you can regenerate them to regain their functionality. Please read our detailed protocol for more information regarding this.
However, we would recommend using Ni-INDIGO products instead. They work with the same buffers and protocols as the Ni-NTA products but have a DTT tolerance of 20 mM.
However, we would recommend using Ni-INDIGO products instead. They work with the same buffers and protocols as the Ni-NTA products but have a DTT tolerance of 20 mM.